|
Not less than once per year, VA will identify a subset of multi-source medications as Tier 1 medications using the criteria below. (D) Tier 3 medication means a medication approved by the FDA under a New Drug Application (NDA) or a biological product approved by the FDA pursuant to a biologics license agreement (BLA) that retains its patent protection and exclusivity and is not a multi-source medication identified in paragraph (b)(1)(iv)(A)( 3) or ( 4) of this section. (C) Tier 2 medication means a multi-source medication that is not identified using the process described in paragraph (b)(2) of this section. (B) Tier 1 medication means a multi-source medication that has been identified using the process described in paragraph (b)(2) of this section. (4) A listed drug, as defined in 21 CFR 314.3, that has been approved under FDCA section 505(c) and is marketed, sold, or distributed directly or indirectly to retail class of trade with either labeling, packaging (other than repackaging as the listed drug in blister packs, unit doses, or similar packaging for use in institutions), product code, labeler code, trade name, or trademark that differs from that of the listed drug. This may include but is not limited to insulin and levothyroxine. (ii) Has the same active ingredient or active ingredients, works in the same way and in a comparable amount of time, and is determined by VA to be substitutable for another medication that has been and remains approved by the FDA pursuant to FDCA section 505(b)(1) or PHSA section 351(a). (i) Has been and remains approved by the FDA pursuant to FDCA section 505(b)(1) or PHSA section 351(a) and (iii) Which is covered by a contracting strategy in place with pricing such that it is lower in cost than other generic sources. (ii) Which is referenced by at least one FDA-approved product that meets the criteria of paragraph (b)(1)(iv)(A)( 1) of this section and (i) Has been and remains approved by the FDA pursuant to FDCA section 505(b)(1) or PHSA section 351(a) FDA determines both therapeutic equivalence for drugs and interchangeability for biological products. 262), and that has been granted an I or B rating in the current version of the FDA's Lists of Licensed Biological Products with Reference Product Exclusivity and Biosimilarity or Interchangeability Evaluations (the Purple Book). (ii) Under section 351(k) of the Public Health Service Act (PHSA, 42 U.S.C. 355), and that has been granted an A-rating in the current version of the FDA's Approved Drug Products with Therapeutic Equivalence Evaluations (the Orange Book) or (i) Under sections 505(b)(2) or 505(j) of the Food, Drug, and Cosmetic Act (FDCA, 21 U.S.C. (1) A medication that has been and remains approved by the FDA. (A) Multi-source medication is any one of the following: 
(iii) For each 30-day or less supply of Tier 3 medications, the copayment amount is $11. (ii) For each 30-day or less supply of Tier 2 medications, the copayment amount is $8. 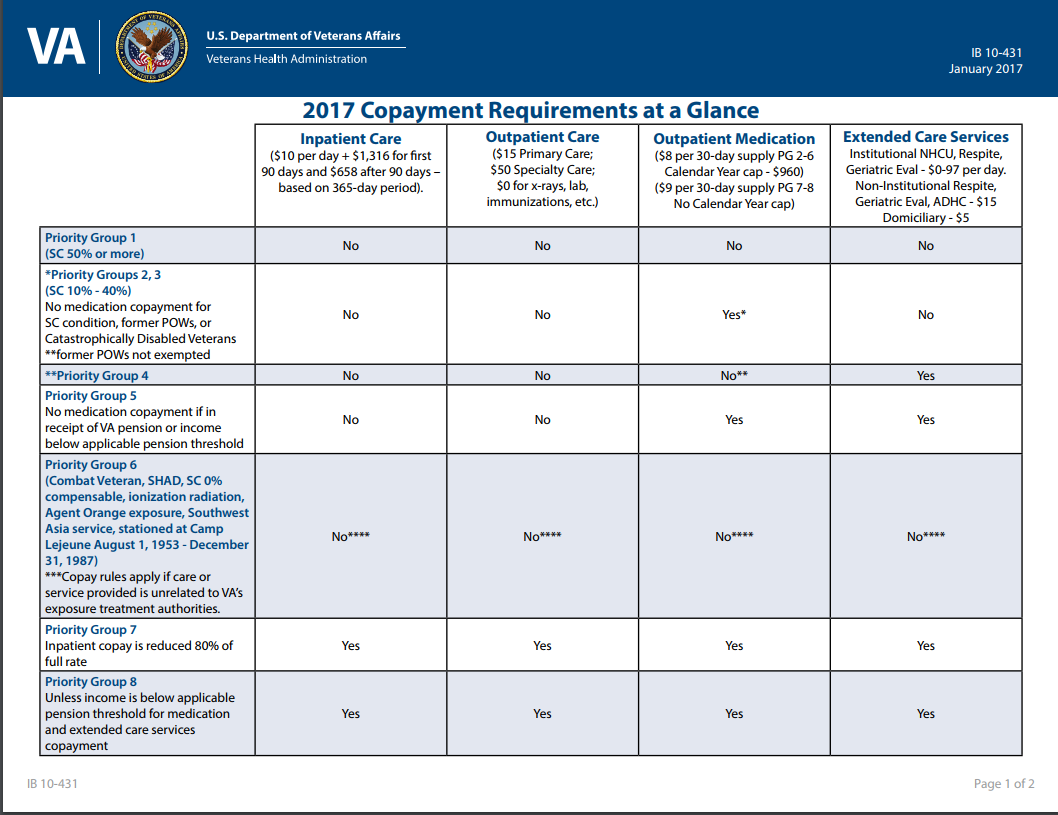
(i) For each 30-day or less supply of Tier 1 medications, the copayment amount is $5. Unless exempted under paragraph (c) of this section, a veteran is obligated to pay VA a copayment for each 30-day or less supply of medication provided by VA on an outpatient basis (other than medication administered during treatment). 
Oral nutritional supplements are commercially prepared nutritionally enhanced products used to supplement the intake of individuals who cannot meet nutrient needs by diet alone. For purposes of this section, the term “medication” means prescription and over-the-counter medications, as determined by the Food and Drug Administration (FDA), but does not mean medical supplies, oral nutritional supplements, or medical devices. This section sets forth requirements regarding copayments for medications provided to veterans by VA.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
